Updated December, 2025
Battery research is evolving quickly as manufacturers explore new chemistries, electrode designs, and fabrication methods. Yet one challenge remains constant: how to transform the flood of experimental data into actionable insights. From Li⁺ diffusion constants to metrology files and electrochemical tests, the process of linking materials research to cell performance is often slow, fragmented, and error-prone.
Smart battery analytics offers a solution. By threading data across R&D workflows, automating calculations, and unifying results into a single platform, researchers can accelerate discovery, reduce costs, and gain a competitive edge.
The Challenges of Cell and Material Innovation
Battery scientists are testing a wide variety of materials approaches to improve lithium-ion batteries (LIBs) and alternative chemistries. Examples include:
-
Uniform cathode materials to enable faster charge/discharge rates
-
SiO and SiC anodes to boost conductivity and cycling ability
-
Solid-state electrolytes and flexible separators
While promising, these innovations introduce complexity. At every stage—materials synthesis, metrology, cell fabrication, and testing—large datasets are generated. For example:
-
Material synthesis yields inputs like milling time, temperature, and atmosphere.
-
Characterization adds SEM images, particle size distribution (PSD), and NMR results.
-
Cell fabrication requires slurry composition, press force, and assembly variables.
-
Testing produces electrochemical profiles, CV, GITT, and impedance data.
Traditionally, teams manage this by creating spreadsheets to track materials as they move from slurries into electrodes and finally into cells. However, this data threading problem consumes enormous time and makes it difficult to relate performance back to material properties.
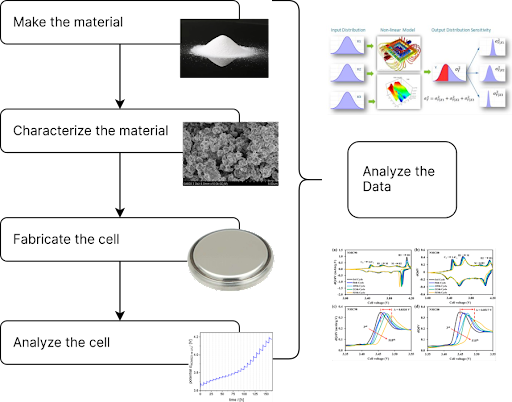
How Smart Battery Analytics Solve the Problem
Automating Key Measurements
Take Li⁺ diffusion constant measurements as an example. These values, critical for understanding kinetics, often fall in the 10⁻¹¹ to 10⁻¹³ cm²·s⁻¹ range. Techniques such as GITT, cyclic voltammetry, or impedance spectroscopy can capture the data, but most cyclers do not analyze it automatically.
With smart battery analytics, platforms like Peaxy Lifecycle Intelligence (PLI) provide scripts that calculate diffusion constants directly, streamlining analysis and eliminating manual effort.
Streamlining Metrology and Image Analysis
Metrology files—SEM, FTIR, BET, or PSD—can be ingested automatically into a smart analytics system. Instead of relying on desktop tools like ImageJ or Fiji, cloud-based scripts process images to calculate particle sizes, detect defects, and refine XRD plots.
As a result, researchers save time and reduce variability in results. Moreover, a hierarchical serial number system ensures that each material, cell, and test remains linked in a unified digital thread.
Accelerating R&D and Reducing Costs
The power of smart battery analytics lies in connecting all four stages of the R&D workflow:
-
Material creation – capture conditions and components.
-
Characterization – process and store metrology results.
-
Cell fabrication – track slurry, electrode, and assembly details.
-
Cell analysis – integrate test data with earlier steps.
By threading these steps together, PLI helps organizations:
-
Shorten the time from material design to validation.
-
Predict outcomes of material changes, reducing trial-and-error.
-
Lower costs by eliminating redundant testing.
-
Gain insights into optimal compositions for performance, longevity, and safety.
This approach applies not only to lithium-ion batteries but also to emerging chemistries, making it a future-proof strategy for R&D organizations.
Key Takeaways
-
Smart battery analytics provides a data-driven framework for accelerating material and cell innovation.
-
Automated scripts reduce the manual burden of diffusion constant calculations and image analysis.
-
Threading data across R&D stages ensures traceability, efficiency, and accuracy.
-
Platforms like PLI help researchers cut costs while pushing new materials and cells to market faster.
FAQ: Smart Battery Analytics and Cell Innovation
Q1. What are smart battery analytics?
Smart battery analytics are digital tools that integrate, automate, and analyze data across the battery R&D workflow. They connect material synthesis, cell fabrication, metrology, and performance testing into a single digital thread.
Q2. How do smart battery analytics accelerate material innovation?
They reduce time spent aggregating and interpreting data by automating calculations (e.g., Li⁺ diffusion constants) and linking results directly to material properties, enabling faster discovery.
Q3. Why is Li⁺ diffusion constant important?
The Li⁺ diffusion constant strongly influences charge and discharge performance. Smart analytics make calculating this constant easier and more reliable.
Q4. Can smart battery analytics reduce R&D costs?
Yes. By predicting outcomes of material changes, they minimize trial-and-error experiments and streamline workflows, cutting both time and cost.
Q5. Are smart battery analytics limited to lithium-ion batteries?
No. While widely applied to LIBs, smart analytics are adaptable to other chemistries, including solid-state and sodium-ion batteries.








